1.A.58 The Type B Influenza Virus Matrix Protein 2 (BM2-C) Family
The mechanisms and functions of viral channel proteins have been reviewed by Fischer and Hsu (2011), Fischer et al. (2012), Nieto-Torres et al. 2015, and Hyser and Estes 2015. Successful uncoating of the influenza B virus in endosomes is predicted to require acidification of the interior of the virus particle. The BM2 integral membrane protein, when expressed in Xenopus oocytes or in mammalian cells, causes acidification of the cells and possesses ion channel activity consistent with proton conduction (Mould et al., 2003). Overexpression of BM2 causes a delay in intracellular transport in the exocytic pathway and causes morphological changes in the Golgi by equilibrating the pH gradient between the Golgi and the cytoplasm. The transmembrane domain of BM2 protein and the influenza A virus A/M2 ion channel proteins both contain the motif HXXXW, and, for both proteins, the His and Trp residues are important for channel function (Mould et al., 2003). 45 residue regions of the two proteins align with 20% identity, 45% similarity and no gaps. Pore-lining residues have been indentified and a model has been proposed (Ma et al., 2008).
BM2 forms a low-pH-gated proton-selective channel. It also transports K+ (Hyser and Estes 2015). Trp-23 may function as a minimalistic gate that opens and closes the pore. When the environmental pH is lower than a threshold, the BM2 channel activates and selectively transports protons across the membrane from the extracellular side to the cytoplasmic side, crucial for the uncoating process. When the virion is internalized into the endosome, the channel acidifies the virion's interior, promoting the dissociation of matrix protein 1 (M1) from the ribonucleoprotein (RNP) thus allowing the transport of the RNP from the virion into the cell's nucleus. It also plays a role in the viral protein secretory pathway. It elevates the intravesicular pH of normally acidic compartments, such as trans-Golgi network, plays a crucial role in virion assembly and is expressed in the late phase of the infection. The cytoplasmic domain of the BM2 protein is required for firm association of the M1 protein with lipid membranes, vRNP complex incorporation into virions and virion morphology (Imai et al., 2008).
The influenza B virus BM2 protein contains 109 amino acid residues and is translated from a bicistronic mRNA in an open reading frame that is +2 nucleotides with respect to the matrix (M1) protein. The amino acid sequence of BM2 contains a hydrophobic region (residues 7-25) that could act as a transmembrane (TM) anchor. BM2 is one of four integral membrane protein encoded by influenza B virus in addition to hemagglutinin (HA), neuraminidase (NA), and the NB glycoprotein. BM2 adopts an N(out) C(in) orientation in membranes and is expressed at the cell surface (Paterson et al., 2003). As the BM2 protein possesses only a single hydrophobic domain and lacks a cleavable signal sequence, it resembles the M(2), NB, and CM2 proteins of influenza A, B, and C viruses, respectively. Chemical cross-linking studies, subunit mixing experiments and fluorescence resonance energy transfer (FRET) analyses all suggest a tetrameric structure (Balannik et al., 2007). Further, the transmembrane domain is largely responsible for oligomerization. Comparison of the amino acid sequence of the TM domain of the BM2 protein with the sequence of the TM domain of the proton-selective ion channel M(2) protein of influenza A virus is intriguing as M(2) protein residues critical for ion selectivity/activation and channel gating (H(37) and W(41), respectively) are found at the same relative position and spacing in the BM2 protein (H(19) and W(23)) (Paterson et al., 2003).
The high resolution conformation of BM2(1-51) in phospholipid bilayers at high pH, corresponding to the closed state of the channel, has been determined using 2D and 3D correlation NMR. Residues 6-28 form a well-ordered alpha-helix, whereas residues 1-5 and 29-35 are probably random coil or beta-sheet conformations. The length of the BM2-TMS helix resembles that of the AM2-TMS, despite their markedly different amino acid sequences (Mandala et al. 2019). The BM2 proton channel is activated by acidic pH to mediate virus uncoating. Unlike influenza A M2 (AM2), which conducts protons with strong inward rectification, BM2 conducts protons, both inwardly and outwardly. Mandala et al. 2020 reported 1.4- and 1.5-Å solid-state NMR structures of the transmembrane domain of the closed and open BM2 channels in a phospholipid environment. Upon activation, the transmembrane helices increase the tilt angle by 6 degrees, and the average pore diameter enlarges by 2.1 Å. BM2 thus undergoes a scissor motion for activation, which differs from the alternating-access motion of AM2. Thus, asymmetric proton conduction requires a backbone hinge motion, whereas bidirectional conduction is achieved by a symmetric scissor motion. The proton-selective histidine and gating tryptophan in the open BM2 reorient on the microsecond timescale, similar to AM2, indicating that side chain dynamics are the essential driver of proton shuttling (Mandala et al. 2020). The membrane environment is an important factor influencing the conformation and hydration of BM2 (Zhang et al. 2020).
The generalized transport reaction catalyzed by BM2 is:
ions including H+ (out) 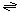 ions including H+ (in).
ions including H+ (in).