1.C.35 The Amoebapore (Amoebapore) Family
Amoebapores are protein capable of forming ion channels (pores) in lipid membranes. They are synthesized by the human protozoan parasite, Entamoeba histolytica, the causative agent of amoebiasis. The major pathogenic effect of E. histolytica is its cytolytic capability which is due to amoebapore which kills target cells. Three isoforms of amoebapore have been isolated and characterized biochemically. The mature proteins are 77-88 amino acids in length, although the precursor proteins are larger. Active peptides are present inside cytoplasmic granules of the amoeba trophozoite. Release allows amoebapore to be inserted into the membrane of a target cell without dependency on a specific receptor. The inserted amoebapore protein oligomerizes to form ion channels which lyse the cell. Distant homologues are found in mammals and slime molds. For example, NK-lysin is an effector peptide of cytolytic T-cells and natural killer (NK) cells. Its synthesis is induced by interleukin 2, and it exhibits antibacterial, antifungal, and antitumor activities.
The amoebapore superfamily includes in addition to the amoebapores (1) the saposins, (2) the NK-lysins and granulysins, (3) the pulmonary surfactant proteins B, (4) the acid sphingomyelinases, (5) acyloxyacyl hydrolases and (6) the aspartic proteases. These amoebapore homologues have many properties in common including membrane binding and stability. A new protein, countin, from the cellular slime mold, Dictyostelium discoideum, comprises the eighth family within this superfamily. Most of these other families includes proteins that have not been shown to function in pore-formation.
The generalized transport reaction catalyzed by amoebapores and their homologues is:
Small molecules (in) 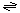 Small molecules (out)
Small molecules (out)