| 1.A.19 The Type A Influenza Virus Matrix-2 Channel (M2-C) Family
The mechanisms and functions of viral channel proteins have been reviewed by Fischer and Hsu (2011) and Fischer et al. (2012). The M1 and M2 matrix proteins of influenza virus type A are produced by alternative splicing of RNA segment 7, but only the first 9 residues are shared by the two proteins. The M2 'matrix' protein is a 97 amino acyl residue long integral membrane protein with an N-terminal extracellular domain (residues 1-23), a single transmembrane domain (residues 24-44) [D24PLVVAASIIGILHLILWILD44] and a large cytoplasmic domain (residues 45-97). It associates into pseudosymmetric homotetramers of parallel transmembrane α-helices with a tilt angle of 25-32o; (Tian et al., 2002). These four hydrophobic α-helices form the proton-selective channel. A 25-residue synthetic peptide of the same sequence forms a H+-selective channel with properties similar to those of the native 97-residue protein. Detailed structural information regarding the transmembrane domain of M2 is available. A two tetramer structure has been solved at 2.1 Å resolution with and without the inhibitor, amantadine. M2 resembles the NB glycopeptide ion channel of influenza virus type B (TC #1.A.32) in size, topology and function but lacks sequence similarity with it (Pielak and Chou, 2010). Not only the character of the membrane environment, but also the abundance of the phospholipid environment is important to achieve the M2 native structure (Wright et al. 2022). Large-scale reorientations of transmembrane helices during Influenza A M2 channel opening have been described (Paschke et al. 2023). The close state of influenza A M2 transmembrane domain is responsive to subtle structural changes of adamantyl amines (Georgiou et al. 2023).
The M2 channel protein is an essential component of the viral envelope because of its ability to form a highly selective, pH-regulated, proton-conducting channel. The virus enters the cell by internalization via the endocytic pathway. Viral uncoating, facilitated by the M2 H+ channel, takes place in the endosomes. The M2 channel allows protons to enter the virus' interior, and acidification weakens the interaction of the M1 protein with the ribonuclear core. M2 also modulates the pH of the trans-Golgi network. The anti-influenza virus drug, amantadine, is a specific blocker of the M2 H+ channel. In the presence of amantadine, viral uncoating is incomplete, and the ribonucleoprotein core fails to promote infection. Aminoadamantanes, including amantadine and rimantadine have been widely
abandoned due to virus resistance, but thermodynamic considerations allow design of new derivatives (Homeyer et al. 2015). M2 exhibits 107 x selectivity for H+ over K+ (Moffat et al., 2008). ~250 H+ are transported per second, and the pKm is about 4.7 (Ivanovic et al., 2012).
Mechanistic analyses of the M2 channel have provided evidence against a mechanism involving the flux of hydronium ions (H3O+) through a channel. Instead, H+ is believed to interact with titratable histidyl groups within the channel. In this mechanism, two histidines in two separate subunits exist, one protonated (His+), the other unprotonated (His0), and the channel is open to the outside. When His0 binds H+, electrostatic repulsion causes a conformational change in the channel so that it opens inwardly. When the H+ passes in, the conformation reverts. This 'carrier-type' mechanism is dependent on the lipid environment and is consistent with the slow rate observed for H+ transport (Cady et al., 2007; Duong-Ly et al., 2005). The channel is also capable of transporting NH4+ and other cations. The M2 amphipathic helices facilitate pH-dependent conformational transition in influenza A virus (Torabifard et al. 2020).
Acharya et al., (2010) suggested that the conduction mechanism involves the exchange of protons between the His37 imidazole moieties of M2 and waters confined to the M2 bundle interior. Protons are conducted as the total charge of the four His37 side chains passes through 2(+) and 3(+) with a pKa near 6. A 1.65 A resolution X-ray structure of the transmembrane protein (residues 25-46), crystallized at pH 6.5, reveals a pore that is lined by alternating layers of sidechains and well-ordered water clusters, which offer a pathway for proton conduction. The His37 residues form a box-like structure, bounded on either side by water clusters with well-ordered oxygen atoms near by. The conformation of the protein, which is intermediate between structures previously solved at higher and lower pH, suggests a mechanism by which conformational changes might facilitate asymmetric diffusion through the channel in the presence of a proton gradient. Moreover, protons diffusing through the channel need not be localized to a single His37 imidazole, but instead may be delocalized over the entire His-box and associated water clusters. Thus, the new crystal structure provides a possible unification of the discrete site versus continuum conduction models (Acharya et al., 2010).
Leiding et al. (2010) showed that M2 has antiporter-like activity, facilitating K+ or Na+ efflux when protons flow down a concentration gradient into the vesicles. Cation efflux is very small except under conditions mimicking those encountered by the endosomally entrapped virus, in which protons are flowing through the channel. This proton/cation exchange function is consistent with the known high proton selectivity of the channel. Thus, M2 acts as a proton uniporter that occasionally allows K+ to flow to maintain electrical neutrality. Remarkably, as the pH inside M2-containing vesicles (pH(in)) decreases, the proton channel activity of M2 is inhibited, but its cation transport activity is activated. This reciprocal inhibition of proton flux and activation of cation flux with decreasing pH(in) first allows accumulation of protons in the early stages of acidification, then trapping of protons within the virus when low pH(in) is achieved.
The indole moiety of the single transmembrane tryptophan residue (position 41) is responsible for H+ gating (Tang et al., 2002). Thus, the side chain of Trp41 probably blocks the pore when the pHout is high so it is closed. When the pH is low, this side chain leaves the pore so it is open.The determinants for folding, drug binding, and proton translocation are packaged in a remarkably small peptide; residues 22-46 in M2 of 97aas (Ma et al., 2009).
Stouffer et al. (2008) described the crystal structure of the transmembrane-spanning region of the homotetrameric M2 protein in the presence and absence of the channel-blocking drug amantadine. pH-dependent structural changes occur near a set of conserved His and Trp residues that are involved in proton gating. The drug-binding site is lined by residues that are mutated in amantadine-resistant viruses. Binding of amantadine physically occludes the pore, and might also perturb the pK(a) of the critical His residue. A multistep mechanism allows the protein to fine-tune its pH-rate profile over a wide range of proton concentrations, arising from different protonation states of the H37 tetrad (Balannik et al., 2010).
In addition to its role in release of viral nucleoproteins, M2 in the trans-Golgi network (TGN) membrane prevents premature conformational rearrangement of newly synthesized haemagglutinin during transport to the cell surface by equilibrating the pH of the TGN with that of the host cell cytoplasm. Inhibiting the proton conductance of M2 using the anti-viral drug amantadine or rimantadine inhibits viral replication. The structure of the tetrameric M2 channel in complex with rimantadine has also been determined by NMR (Schnell and Chou, 2008). In the closed state, four tightly packed transmembrane helices define a narrow channel, in which a 'tryptophan gate' is locked by intermolecular interactions with aspartic acid. A carboxy-terminal, amphipathic helix oriented nearly perpendicular to the transmembrane helix forms an inward-facing base. Lowering the pH destabilizes the transmembrane helical packing and unlocks the gate, admitting water to conduct protons, whereas the C-terminal base remains intact, preventing dissociation of the tetramer. Rimantadine binds at four equivalent sites near the gate on the lipid-facing side of the channel and stabilizes the closed conformation of the pore. Drug-resistance mutations are predicted to counter the effect of drug binding by either increasing the hydrophilicity of the pore or weakening helix-helix packing, thus facilitating channel opening. Amantadine binds to the pore (Jing et al., 2008). The M2 amphipathic helix causes membrane curvature, which in turn perturbs the TM helix conformation (Wang et al., 2012).
The role of histidine in channel-forming transmembrane (TM) helices was investigated by comparing the TM helices from Virus protein 'u' (Vpu) and the M2 proton channel. Both proteins are members of the viroporin family of small membrane proteins that exhibit ion channel activity, and have a single TM helix that is capable of forming oligomers. The TM helices from both proteins have a conserved tryptophan towards the C-terminus. Alanine 18 of Vpu has been mutated to histidine in order to artificially introduce the same HXXXW motif that is central to the proton channel activity of M2. The mutated Vpu TMS resulted in an increase in helix tilt angle of 11° in lipid bilayers compared to the wild-type Vpu TMS. Wang et al. (2013) found the reverse when histidine 37 of the HXXXW motif in M2 was mutated to alanine; it decreased the helix tilt by 10° from that of wild-type M2. The tilt change was independent of both the helix length and the presence of tryptophan. Compared to wild-type M2, the H37A mutant displayed a lowered sensitivity to the proton concentration. The solvent accessibility of histidine-containing M2 was greater than without histidine. This suggests that the TM helix may increase the solvent exposure by changing its tilt angle in order to accommodate a polar/charged residue within the hydrophobic membrane region. The comparative results of M2, Vpu and their mutants demonstrate the significance of histidine in a transmembrane helix and the plasticity of the function and structure of ion channels stemming from changes at a single amino acid site.
Water molecules within the
pore form hydrogen-bonded networks or 'water wires' from the channel entrance to
His37. Pore-lining carbonyl groups are well situated to stabilize hydronium ions via second-shell
interactions involving bridging water molecules. A
collective switch of hydrogen bond orientations may contribute to the directionality of proton
flux as His37 is dynamically protonated and deprotonated in the conduction cycle (Thomaston et al. 2015). M2 drug inhibitors interact with and disrupt networks of hydrogen-bonded waters that are widely utilized throughout nature to facilitate proton diffusion within proteins (Thomaston et al. 2018).
M2 colocalized and interacted with MAVS (mitochondrial antiviral signaling protein) on mitochondria, and positively regulated MAVS-mediated innate immunity. It induces reactive oxygen species (ROS) production that is required for activation of macroautophagy/autophagy and enhancement of MAVS signaling. The proton channel activity of M2 protein is essential for ROS production and antagonizing the autophagy pathway to control MAVS aggregation, thereby enhancing MAVS signal activity. Thus, M2 modulates host antiviral immunity (Wang et al. 2019).
Diazabicyclooctane derivatives with a constant charge of +2 block proton diffusion through the M2 ion channel (Vorobjev 2020). Two types of diazabicyclooctane derivatives were analyzed for binding to the M2 channel. An optimal structure was determined for a blocker to most efficiently block proton diffusion. The new molecule is advantageous over amantadine and rimantadine in having a positive charge of +2, which creates a positive electrostatic potential barrier to proton transport in addition to a steric barrier.
The generalized transport reaction catalyzed by the M2 channel is:
H+ (out) 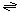 H+ (in). H+ (in).
|
This family belongs to the Influenza A/B Virus M2 Protein (M2) Superfamily.
|
| References: |
Acharya, R., V. Carnevale, G. Fiorin, B.G. Levine, A.L. Polishchuk, V. Balannik, I. Samish, R.A. Lamb, L.H. Pinto, W.F. DeGrado, and M.L. Klein. (2010). Structure and mechanism of proton transport through the transmembrane tetrameric M2 protein bundle of the influenza A virus. Proc. Natl. Acad. Sci. USA 107: 15075-15080.
|
Andreas, L.B., M. Reese, M.T. Eddy, V. Gelev, Q.Z. Ni, E.A. Miller, L. Emsley, G. Pintacuda, J.J. Chou, and R.G. Griffin. (2015). Structure and Mechanism of the Influenza A M218-60 Dimer of Dimers. J. Am. Chem. Soc. 137: 14877-14886.
|
Balannik, V., V. Carnevale, G. Fiorin, B.G. Levine, R.A. Lamb, M.L. Klein, W.F. Degrado, and L.H. Pinto. (2010). Functional studies and modeling of pore-lining residue mutants of the influenza a virus M2 ion channel. Biochemistry 49: 696-708.
|
Cady, S.D., C. Goodman, C.D. Tatko, W.F. DeGrado, and M. Hong. (2007). Determining the orientation of uniaxially rotating membrane proteins using unoriented samples: a 2H, 13C, and 15N solid-state NMR investigation of the dynamics and orientation of a transmembrane helical bundle. J. Am. Chem. Soc. 129: 5719-5729.
|
Cristian, L., J.D. Lear, and W.F. DeGrado. (2003). Use of thiol-disulfide equilibria to measure the energetics of assembly of transmembrane helices in phospholipid bilayers. Proc. Natl. Acad. Sci. USA 100: 14772-14777.
|
Duong-Ly, K.C., V. Nanda, W.F. Degrado, and K.P. Howard. (2005). The conformation of the pore region of the M2 proton channel depends on lipid bilayer environment. Protein Sci. 14: 856-861.
|
Elkins, M.R., J.K. Williams, M.D. Gelenter, P. Dai, B. Kwon, I.V. Sergeyev, B.L. Pentelute, and M. Hong. (2017). Cholesterol-binding site of the influenza M2 protein in lipid bilayers from solid-state NMR. Proc. Natl. Acad. Sci. USA 114: 12946-12951.
|
Fischer, W.B. and H.J. Hsu. (2011). Viral channel forming proteins - modeling the target. Biochim. Biophys. Acta. 1808: 561-571.
|
Fischer, W.B. and M.S. Sansom. (2002). Viral ion channels: structure and function. Biochim. Biophys. Acta 1561: 27-45.
|
Fischer, W.B., M. Pitkeathly, B.A. Wallace, L.R. Forrest, G.R. Smith, and M.S.P. Sansom. (2000). Transmembrane peptide NB of influenza B: a simulation, structure and conductance study. Biochemistry 41: 12708-12716.
|
Fischer, W.B., Y.T. Wang, C. Schindler, and C.P. Chen. (2012). Mechanism of function of viral channel proteins and implications for drug development. Int Rev Cell Mol Biol 294: 259-321.
|
Georgiou, K., A. Konstantinidi, J. Hutterer, K. Freudenberger, F. Kolarov, G. Lambrinidis, I. Stylianakis, M. Stampelou, G. Gauglitz, and A. Kolocouris. (2023). Accurate calculation of affinity changes to the close state of influenza A M2 transmembrane domain in response to subtle structural changes of adamantyl amines using free energy perturbation methods in different lipid bilayers. Biochim. Biophys. Acta. Biomembr 1866: 184258. [Epub: Ahead of Print]
|
Homeyer, N., H. Ioannidis, F. Kolarov, G. Gauglitz, C. Zikos, A. Kolocouris, and H. Gohlke. (2015). Interpreting thermodynamic profiles of aminoadamantane compounds inhibiting the M2 proton channel of influenza A by free energy calculations. J Chem Inf Model. [Epub: Ahead of Print]
|
Hong M. and DeGrado WF. (2012). Structural basis for proton conduction and inhibition by the influenza M2 protein. Protein Sci. 21(11):1620-33.
|
Ivanovic, T., R. Rozendaal, D.L. Floyd, M. Popovic, A.M. van Oijen, and S.C. Harrison. (2012). Kinetics of proton transport into influenza virions by the viral m2 channel. PLoS One 7: e31566.
|
Jing, X., C. Ma, Y. Ohigashi, F.A. Oliveira, T.S. Jardetzky, L.H. Pinto, and R.A. Lamb. (2008). Functional studies indicate amantadine binds to the pore of the influenza A virus M2 proton-selective ion channel. Proc. Natl. Acad. Sci. USA 105: 10967-10972.
|
Kalita, M.M. and W.B. Fischer. (2017). Decoupled side chain and backbone dynamics for proton translocation - M2 of influenza A. J Mol Model 23: 212.
|
Kim, G., H.E. Raymond, A.L. Herneisen, A. Wong-Rolle, and K.P. Howard. (2019). The distal cytoplasmic tail of the influenza A M2 protein dynamically extends from the membrane. Biochim. Biophys. Acta. Biomembr 1861: 1421-1427.
|
Kolokouris, D., I.E. Kalenderoglou, A.L. Duncan, R.A. Corey, M.S.P. Sansom, and A. Kolocouris. (2025). The Role of Cholesterol in M2 Clustering and Viral Budding Explained. J Chem Theory Comput 21: 912-932.
|
Kordyukova, L.V., P.V. Konarev, N.V. Fedorova, E.V. Shtykova, A.L. Ksenofontov, N.A. Loshkarev, L.A. Dadinova, T.A. Timofeeva, S.S. Abramchuk, A.V. Moiseenko, L.A. Baratova, D.I. Svergun, and O.V. Batishchev. (2021). The Cytoplasmic Tail of Influenza A Virus Hemagglutinin and Membrane Lipid Composition Change the Mode of M1 Protein Association with the Lipid Bilayer. Membranes (Basel) 11:.
|
Kukol, A., P.D. Adams, L.M. Rice, A.T. Brunger, and I.T. Arkin. (1999). Experimentally based orientational refinement of membrane protein models: a structure for the influenza A M2 H+ channel. J. Mol. Biol. 286: 951-962.
|
Kumar, U., P. Goyal, Z.K. Madni, K. Kamble, V. Gaur, M.S. Rajala, and D.M. Salunke. (2023). A structure and knowledge-based combinatorial approach to engineering universal scFv antibodies against influenza M2 protein. J Biomed Sci 30: 56.
|
Leiding, T., J. Wang, J. Martinsson, W.F. DeGrado, and S.P. Arsköld. (2010). Proton and cation transport activity of the M2 proton channel from influenza A virus. Proc. Natl. Acad. Sci. USA 107: 15409-15414.
|
Liang, R., J.M. Swanson, J.J. Madsen, M. Hong, W.F. DeGrado, and G.A. Voth. (2016). Acid activation mechanism of the influenza A M2 proton channel. Proc. Natl. Acad. Sci. USA. [Epub: Ahead of Print]
|
Liao, S.Y., Y. Yang, D. Tietze, and M. Hong. (2015). The influenza m2 cytoplasmic tail changes the proton-exchange equilibria and the backbone conformation of the transmembrane histidine residue to facilitate proton conduction. J. Am. Chem. Soc. 137: 6067-6077.
|
Ma, C., A.L. Polishchuk, Y. Ohigashi, A.L. Stouffer, A. Schön, E. Magavern, X. Jing, J.D. Lear, E. Freire, R.A. Lamb, W.F. DeGrado, and L.H. Pinto. (2009). Identification of the functional core of the influenza A virus A/M2 proton-selective ion channel. Proc. Natl. Acad. Sci. USA 106: 12283-12288.
|
Mandala, V.S., M.D. Gelenter, and M. Hong. (2018). Transport-Relevant Protein Conformational Dynamics and Water Dynamics on Multiple Time Scales in an Archetypal Proton Channel: Insights from Solid-State NMR. J. Am. Chem. Soc. 140: 1514-1524.
|
Moffat, J.C., V. Vijayvergiya, P.F. Gao, T.A. Cross, D.J. Woodbury, and D.D. Busath. (2008). Proton transport through influenza A virus M2 protein reconstituted in vesicles. Biophys. J. 94: 434-445.
|
Mould, J.A., H. Li, C.S. Dudlak, J.D. Lear, A. Pekosz, R.A. Lamb, and L.H. Pinto. (2000). Mechanism for proton conduction of the M2 ion channel of influenza A virus. J. Biol. Chem. 275: 8592-8599.
|
Mould, J.A., J.E. Drury, S.M. Frings, U.B. Kaupp, A. Pekosz, R.A. Lamb, and L.H. Pinto. (2000). Permeation and activation of the M2 ion channel of influenza A virus. J. Biol. Chem. 75: 31038-31050.
|
Musharrafieh, R., P. Lagarias, C. Ma, G. Tan, A. Kolocouris, and J. Wang. (2019). The L46P mutant confers a novel allosteric mechanism of resistance towards the influenza A virus M2 S31N proton channel blockers. Mol Pharmacol. [Epub: Ahead of Print]
|
Paschke, R.R., S. Mohr, S. Lange, A. Lange, and J. Kozuch. (2023). In Situ Spectroscopic Detection of Large-Scale Reorientations of Transmembrane Helices During Influenza A M2 Channel Opening. Angew Chem Int Ed Engl e202309069. [Epub: Ahead of Print]
|
Pielak, R.M. and J.J. Chou. (2010). Flu channel drug resistance: a tale of two sites. Protein Cell 1: 246-258.
|
Pinto, L.H., G.R. Dieckmann, C.S. Gandhi, C.G. Papworth, J. Braman, M.A. Shaughnessy. J.D. Lear, R.A. Lamb, and W.F. DeGrado. (1997). A functionally defined model for the M2 proton channel of influenza A virus suggests a mechanism for its ion selectivity. Proc. Natl. Acad. Sci. USA 94: 11301-11306.
|
Schnell, J.R., and J.J. Chou (2008). Structure and mechanism of the M2 proton channel of influenza A virus. Nature 451: 591-5.
|
Schroeder, C., H. Heider, E. Möncke-Buchner, and T.I. Lin. (2005). The influenza virus ion channel and maturation cofactor M2 is a cholesterol-binding protein. Eur Biophys. J. 34: 52-66.
|
Stouffer, A.L., R. Acharya, D. Salom, A.S. Levine, L. Di Costanzo, C.S. Soto, V. Tereshko, V. Nanda, S. Stayrook, and W.F. DeGrado (2008). Structural basis for the function and inhibition of an influenza virus proton channel. Nature 451: 596-9.
|
Tang, Y., F. Zaitseva, R.A. Lamb, and L.H. Pinto. (2002). The gate of the influenza virus M2 proton channel is formed by a single tryptophan residue. J. Biol. Chem. 277: 39880-39886.
|
Thomaston, J.L., M. Alfonso-Prieto, R.A. Woldeyes, J.S. Fraser, M.L. Klein, G. Fiorin, and W.F. DeGrado. (2015). High-resolution structures of the M2 channel from influenza A virus reveal dynamic pathways for proton stabilization and transduction. Proc. Natl. Acad. Sci. USA 112: 14260-14265.
|
Thomaston, J.L., N.F. Polizzi, A. Konstantinidi, J. Wang, A. Kolocouris, and W.F. DeGrado. (2018). Inhibitors of the M2 Proton Channel Engage and Disrupt Transmembrane Networks of Hydrogen-Bonded Waters. J. Am. Chem. Soc. [Epub: Ahead of Print]
|
Tian, C. K. Tobler, R.A. Lamb, L.H. Pinto, and T.A. Cross. (2002). Expression and initial structural insights from solid-stage NMR of the M2 proton channel from influenza A virus. Biochemistry 41: 11294-11300.
|
Torabifard, H., A. Panahi, and C.L. Brooks, 3rd. (2020). M2 amphipathic helices facilitate pH-dependent conformational transition in influenza A virus. Proc. Natl. Acad. Sci. USA 117: 3583-3591.
|
Townsend, J.A., H.M. Sanders, A.D. Rolland, C.K. Park, N.C. Horton, J.S. Prell, J. Wang, and M.T. Marty. (2021). Influenza AM2 Channel Oligomerization Is Sensitive to Its Chemical Environment. Anal Chem 93: 16273-16281.
|
Vorobjev, Y.N. (2020). [Design of an Efficient Inhibitor for the Influenza A Virus M2 Ion Channel]. Mol Biol (Mosk) 54: 321-332.
|
Wang, R., Y. Zhu, X. Lin, C. Ren, J. Zhao, F. Wang, X. Gao, R. Xiao, L. Zhao, H. Chen, M. Jin, W. Ma, and H. Zhou. (2019). Influenza M2 protein regulates MAVS-mediated signaling pathway through interacting with MAVS and increasing ROS production. Autophagy 1-19. [Epub: Ahead of Print]
|
Wang, T., S.D. Cady, and M. Hong. (2012). NMR determination of protein partitioning into membrane domains with different curvatures and application to the influenza m2 Peptide. Biophys. J. 102: 787-794.
|
Wang, Y., S.H. Park, Y. Tian, and S.J. Opella. (2013). Impact of histidine residues on the transmembrane helices of viroporins. Mol. Membr. Biol. 30: 360-369.
|
Witter, R., F. Nozirov, U. Sternberg, T.A. Cross, A.S. Ulrich, and R. Fu. (2008). Solid-state 19F NMR spectroscopy reveals that Trp41 participates in the gating mechanism of the M2 proton channel of influenza A virus. J. Am. Chem. Soc. 130: 918-924.
|
Wright, A.K., J. Paulino, and T.A. Cross. (2022). Emulating Membrane Protein Environments─How Much Lipid Is Required for a Native Structure: Influenza S31N M2. J. Am. Chem. Soc. 144: 2137-2148.
|
| Examples: |
| TC# | Name | Organismal Type | Example |
| 1.A.19.1.1 | Matrix protein, M2, an acid activated drug-sensitive proton channel. Transport involves binding to the four His-37s and transfer to water molecules on the inside of the channel (Acharya et al., 2010). Functional properties and structure are known (Hong and Degrado 2012). The cytoplasmic tail facilitates proton conduction (Liao et al. 2015). It is a dimer of dimers (Andreas et al. 2015). The four TMSs flanking the channel lumen
alone seem to determine the proton conduction mechanism (Liang et al. 2016). His-37 forms a planar tetrad in the configuration of the bundle
accepting and translocating the incoming protons from the N terminal
side, exterior of the virus, to the C terminal side, inside the virus (Kalita and Fischer 2017). The cholesterol binding site in M2 that mediates membrane scission in a cholesterol-dependent manner to cause virus budding and release has been identified (Elkins et al. 2017).Transport-related conformational changes coupled to water and H+ movements have been studied (Mandala et al. 2018). The L46P mutant confers a novel allosteric mechanism of resistance towards the influenza A virus M2 S31N proton channel blockers (Musharrafieh et al. 2019). The C-terminal domain of M2 may serve as a sensor that regulates how M2 participates in critical events in the viral infection cycle (Kim et al. 2019). The M2 proton channel protein self-assembles into tetramers that retain the ability to bind to the drug amantadine, and the effects of phospholipid acyl chain length and cholesterol on the peptide association were investigated. Association of the helices depends on the thickness of the bilayer and cholesterol levels present in the phospholipid bilayer. The most favorable folding occurred when there was a good match between the width of the apolar region of the bilayer and the hydrophobic length of the transmembrane helix with tighter association upon inclusion of cholesterol in the lipid bilayer (Cristian et al. 2003). The influenza A M2 homotetrameric channel consists of four transmembrane
(TM) and four amphipathic helices (AHs). This viral proton channel is
suggested to form clusters in the catenoid budding neck areas in
raft-like domains of the plasma membrane, resulting in cell membrane
scission and viral release. holesterol-bridged M2 channels can exert a lateral force on the
surrounding membrane to induce the necessary negative Gaussian curvature
profile, which permits spontaneous scission of the catenoid membrane
neck and leads to viral buds and scission (Kolokouris et al. 2025). | Viruses | M2 of influenza virus type A |
| |
| 1.A.19.1.2 | Matrix protein M2 of 96 aas and 1 TMS. Forms
a proton-selective ion channel that is necessary for the efficient
release of the viral genome during virus entry. After attaching to the
cell surface, the virion enters the cell by endocytosis. Acidification
of the endosome triggers M2 ion channel activity. The influx of protons
into the virion interior is believed to disrupt interactions between the
viral ribonucleoprotein (RNP), matrix protein 1 (M1), and lipid
bilayers, thereby freeing the viral genome from interaction with viral
proteins and enabling RNA segments to migrate to the host cell nucleus,
where influenza virus RNA transcription and replication occur. Also
plays a role in viral proteins secretory pathway. The cytoplasmic tail of Influenza A virus hemagglutinin and membrane lipid composition change the mode of M1 protein association with the lipid bilayer (Kordyukova et al. 2021). Universal scFv antibodies against the influenza M2 protein have been prepaired (Kumar et al. 2023).
| | M2 of Influenza A virus (A/flat-faced bat/Peru) |
| |
| 1.A.19.1.3 | Matrix protein 2, M2, of 80 aas and 1 TMS. The influenza virus ion channel and maturation cofactor M2 is a cholesterol-binding protein and M2 may promote clustering, merger of rafts and the pinching-off (fission) of virus particles (Schroeder et al. 2005). | | M2 of Influenza A virus (A/swine) |
| |
| 1.A.19.1.4 | M2 protein of 95 aas and 1 TMS, AM2. This protein shows 28% identity, 50% similarity and 10% gaps with BM2 (TC# 1.B.58.1.1) with residues 8 - 65 aligning with residues 26 - 81. AM2 forms a range of oligomeric complexes that are strongly influenced by the local chemical environment. Native mass spectrometry of AM2 in nanodiscs with different lipids showed that lipids also affected the oligomeric states of AM2. Finally, nanodiscs uniquely enabled the measurement of amantadine binding stoichiometries to AM2 in the intact lipid bilayer (Townsend et al. 2021). | |
AM2 of Influenza A virus (A/herring gull/Newfoundland) |
| |
| Examples: |
| TC# | Name | Organismal Type | Example |
| 1.A.19.2.1 | Protein of 489 aas with C-terminal region resembling the M2 protein (33% identity with no gaps). | | Protein of Apis mellifera filamentous virus |
| |
 H+ (in).
H+ (in).