| 1.B.32 The Fusobacterial Outer Membrane Porin (FomA) Family FomA is the designation given to a small family of sequence similar outer membrane porins from F. nucleatum. They are trimeric proteins, each subunit exhibiting 14 C-terminal antiparallel transmembrane β- strands connected to an N-terminal periplasmic domain (Puntervoll et al., 2002). An external loop, L6, the largest of the seven such loops, when deleted renders FomA more permeable to antibiotics. L6 probably folds back into the β-barrel, thereby constricting the pore. FomA porins lack significant sequence similarity to other known porins, but they do exhibit similar structural features. They transport solutes non-specifically except for a size limit. Deletion of L6 increases this size limit.
The transport reaction catalyzed by FomA porins is: solute (out) 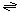 solute (periplasm) solute (periplasm)
|
This family belongs to the Outer Membrane Pore-forming Protein I (OMPP-I) Superfamily .
|
| References: |
Anbazhagan, V., N. Vijay, J.H. Kleinschmidt, and D. Marsh. (2008). Protein-lipid interactions with Fusobacterium nucleatum major outer membrane protein FomA: spin-label EPR and polarized infrared spectroscopy. Biochemistry 47: 8414-8423.
|
Kattner, C., S. Pfennig, P. Massari, and M. Tanabe. (2015). One-step purification and porin transport activity of the major outer membrane proteins P2 from Haemophilus influenzae, FomA from Fusobacterium nucleatum and PorB from Neisseria meningitidis. Appl Biochem Biotechnol 175: 2907-2915.
|
Kleinschmidt, J.H. (2006). Folding kinetics of the outer membrane proteins OmpA and FomA into phospholipid bilayers. Chem Phys Lipids 141: 30-47.
|
Kleivdal, H., P. Puntervoll, and H.B. Jensen. (2001). Topological investigation of the FomA porin from Fusobacterium nucleatum and identification of the constriction loop L6. Microbiology 147: 1059-1067.
|
Kleivdal, H., R. Benz, and H.B. Jensen. (1995). The Fusobacterium nucleatum major outer-membrane protein (FomA) forms trimeric, water-filled channels in lipid bilayer membranes. Eur J Biochem 233: 310-316.
|
Pszon-Bartosz, K., J.S. Hansen, K.B. Stibius, J.S. Groth, J. Emnéus, O. Geschke, and C. Hélix-Nielsen. (2011). Assessing the efficacy of vesicle fusion with planar membrane arrays using a mitochondrial porin as reporter. Biochem. Biophys. Res. Commun. 406: 96-100.
|
Puntervoll, P., H. Kleivdal, K. Ole Dahl, W. Bitter, J. Tommassen, and H.B. Jensen. (2000). The Fusobacterium nucleatum porin FomA possesses the general topology of the non-specific porins. Microbiology 146: 1437-1445.
|
Puntervoll, P., M. Ruud, L.J. Bruseth, H. Kleivdal, B.T. Høgh, R. Benz, and H.B. Jensen. (2002). Structural characterization of the fusobacterial non-specific porin FomA suggests a 14-stranded topology, unlike the classical porins. Microbiology 148: 3395-3403.
|
| Examples: |
| TC# | Name | Organismal Type | Example |
| 1.B.32.1.1 | Non-specific FomA porin precursor (Kleivdal et al. 1995). The N-terminal region is periplasmic while the C-terminus is a 14 stranded β-barrel (Puntervoll et al. 2002). The β-barrel may have a tilt angle of 45° relative to the barrel axis (Anbazhagan et al., 2008). FomA is fusogenic (Pszon-Bartosz et al., 2011). Its unique solute transport activity with size exclusion limit has been described (Kattner et al. 2015). FomA is a voltage-dependent porin, predicted to form a 14 stranded beta-barrel. It folds in a range of model membranes of very different phospholipid compositions. A study on FomA folding into lipid bilayers indicated the presence of parallel folding pathways for OMPs with larger transmembrane beta-barrels (Kleinschmidt 2006). | Fusobacteria | FomA of Fusobacterium nucleatum (Q47905) |
| |
| 1.B.32.1.2 | Putative porin | γ-Proteobacteria | Putative porin of Providencia rustigianii |
| |
| Examples: |
| TC# | Name | Organismal Type | Example |
| 1.B.32.2.1 | Putative porin | Fusobacteria | Putative porin of Ilyobacter polytropus |
| |
| 1.B.32.2.2 | FomA homologue of 294 aas | Fusobacteria | FomA homologue of Ilyobacter polytropus |
| |
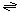 solute (periplasm)
solute (periplasm)