1.A.23 The Small Conductance Mechanosensitive Ion Channel (MscS) Family
The MscS family (the SwissProt UPF0003 family) is a group of topographically diverse proteins, some of which are functionally characterized. They exhibit homology in only a restricted region. Early electrophysiological studies with E. coli suggested the presence of two distinguishable mechanosensitive ion channels, one with large conductance (MscL; TC #1.A.22) and one with small conductance (MscS; this family) (Martinac et al., 1987, 1990). Cytoplasmic beta domains appear to be new gating elements in MscS channels (Koprowski et al., 2011). Open and closed conformational states of the heptameric channel have been proposed and discussed (Pliotas et al. 2012). Evolutionary considerations have been reported (Booth et al. 2015). Some MscS channels may function in amino acid efflux, Ca2+ regulation and cell division (Cox et al. 2015). Mechanosensitive MscS channels can be gated by delipidation (Flegler et al. 2021). Pore lipid modifications modulate MscS nanopore for enhanced single-molecule sensing (Zhao et al. 2025).
Application of a ramp of negative pressure to a patch excised from an E. coli giant spheroplast gave (1) a small conductance (MscS; ~1 nS in 400 mM salt) with a sustained open state, and (2) a large conductance (MscL; ~3 nS) with faster kinetics, activated at higher pressure. MscS was reported to exhibit a weak anionic preference and a voltage dependency, tending to open upon depolarization. Activation by membrane-intercalating amphipathic compounds suggested that the MscS channel is sensitive to mechanical perturbations in the lipid bilayer. It was suggested that MscS plays a role in fast osmoregulatory responses. How these channels may respond to change in the mechanical environment the lipid bilayer provides is discussed by Kung et al. (2010). Mechanosensitive ion channels have been discussed in terms of their physiological importance and their potential roles in cancer (Otero-Sobrino et al. 2023).
Mechanosensitive channels function as electromechanical switches with the capability to sense the physical state of lipid bilayers. The X-ray crystal structures of MscL and MscS allow identification of the types of protein motions associated with the opening and closing of these structurally unrelated channels, while providing the framework to address a mechanism of tension sensing that is defined by channel-lipid interactions (Perozo and Rees, 2003). Functional, structural and dynamic data offer fresh insights into the molecular basis of gating for these membrane proteins. Members of the MscS family are found in bacteria, archaea, fungi, and plants (Wilson et al. 2013). Differential expression of various mscS genes in tissue developmental stages and under stress conditions in plants suggested roles in development and stress responses. Altered expression during CaCl2 stress suggested a role in Ca2+ homeostasis, signaling, growth, and defense responses (Kaur et al. 2020).
Mutations in the genes encoding the KefA (AefA) and YggB proteins of E. coli block the MscS mechanosensitive channel activity. The principal one is affected by knockouts of YggB, while the minor one is affected by knockouts of KefA. These two channels open in response to pressure changes during osmotic downshift just below those that cause cell disruption and death (Biggin and Sansom, 2003; Pivetti et al., 2003). The C-termini of the YggB heptameric channel move apart upon channel opening and may serve as the gate (Koprowski and Kubalski, 2003). High resolution 3-D structures are available (Bass et al., 2002; Lai et al. 2013). Crosslinking studies indicate that a large conformational change accompanies the open to the closed configuration (Miller et al., 2003b).
Homologues of YggB are found in Gram-negative, Gram-positive and cyanobacteria, in archaea, in yeast and in plants, but not in animals (Booth and Louis, 1999). One archaeon, Haloferax volcanii, exhibits mechanosensitive channels similar in conductance and mass to YggB of E. coli, but the sequences of these channel proteins are not available (Le Dain et al., 1998). Two sequenced MscS homologues have been functionally characterized from Methanococcus jannaschii (Kloda and Martinac, 2001a,b).
MscS family homologues vary in length between 248 and 1120 amino acyl residues, but the homologous region that is shared by most of them is only 200-250 residues long, exhibiting 4-5 TMSs (Miller et al., 2003b). The topologies of these proteins differ drastically. YggB (286 aas; spP11666) exhibits 4-5 putative transmembrane α-helical spanners (TMSs); KefA (AefA) (1120 aas; spP77338) exhibits 11 TMSs; YjeP of E. coli (1107 aas; spP39285) exhibits 11 TMSs; YbiO of E. coli (741 aas; spP75783) exhibits 10 TMSs; and YbdG of E. coli (415 aas; spP39455) exhibits 5 TMSs. Moreover, of the M. jannaschii homologues, MJ0170 (350 aas; spQ57634) exhibits 4-5 TMSs; MJ0700 (324 aas; spQ58111) exhibits 4 TMSs; and MJ1143 (361 aas; spQ58543) exhibits 5-6 TMSs. This topological variability is an unusual characteristic of a family of homologous transport proteins, and its functional significance cannot be evaluated at this time. It is possible that transport mechanisms will vary in accordance with topology. On the other hand, only 2 TMSs, common to all of these proteins may comprise the channel (Booth and Louis, 1999).
The E. coli and Synechocystis genomes include five recognized paralogues of the MscS family while the Bacillus subtilis and Methanococcus jannaschii genomes include three paralogues. KefA is multidomain and may be multifunctional. It has a large (470 amino acyl residues) N-terminal extracytoplasmic domain that may interact with the peptidoglycan cell wall, a central hydrophobic region including the 11 TMSs (residues 480-940), and a C-terminal cytoplasmic domain (residues 941-1120). Only the last four TMSs and the C-terminal hydrophilic domain are homologous to the much shorter YggA protein. One of the Synechocystis homologues (slr1575) possesses a C-terminal domain homologous to cyclic AMP-dependent protein kinaseA regulatory subunits (Ochoa de Alde and Houmard, 2000). It may therefore be a cyclic nucleotide-regulated channel.
A homologue of MscS channels in Erwinia chrysanthemi, BspA, is encoded within an operon that includes the psd gene encoding phosphatidyl serine decarboxylase. In high salts medium, glycine betaine initially is taken up normally in a bspA mutant, but uptake is followed by reduced glucose uptake and release of glycine betaine without loss of viability. It was suggested that BspA is not a channel but instead senses the intracellular glycine betaine and the extracellular salt concentrations, and thus serves as a receptor for osmoadaptation (Touzé et al., 2001).
In bacterial and animal systems, mechanosensitive (MS) ion channels are thought to mediate the perception of pressure, touch, and sound. Ten MscS-Like (MSL) proteins are encoded within the genome of Arabidopsis thaliana. MSL2 and MSL3, along with MSC1, a MscS family member from green algae, are implicated in the control of organelle morphology. Haswell et al. (2008) characterized MSL9 and MSL10, two MSL proteins found in the plasma membrane of root cells. MSL9 and MSL10, along with three other members of the MSL family, are required for MS channel activities detected in protoplasts derived from root cells.
The Escherichia coli mechanosensitive channel, MscS, opens to allow rapid ion efflux, relieving the turgor pressure that would otherwise destroy the cell. Wang et al. (2008) described a 3.45 angstrom-resolution structure in an open conformation. MscS has a pore diameter of ~13 angstroms created by substantial rotational rearrangement of the three transmembrane helices. The structure suggests a molecular mechanism that underlies MscS gating and its decay of conductivity during prolonged activation (Wang et al., 2008). The levels of both MscL and MscS channels in Bacillus subtilis are high during exponential phase growth, very low in stationary phase and non-detectable in spores (Wahome et al., 2009).
The E. coli MscS (EcMscS) has been extensively studied, but it may display characteristics not widely conserved in this protein family. With numerous members now electrophysiologically characterized, these channels displays a breadth of ion selectivity with both anion and cation selective members. The selectivities of these channels may be relatively weak in comparison to voltage-gated channels. Residues important for selectivity in MscS homologs suggest different selectivity mechanisms than those employed by voltage gated K+, Na+, Ca2+ and Cl- channels whose selectivity filters are housed within their transmembrane pores. Cox et al. 2013 attempted to unravel the potential physiological relevance of these differences.
Mechanosensitive (MS) channels provide protection against hypo-osmotic shock in bacteria whereas eukaryotic MS channels fulfill a multitude of important functions in addition to osmoregulation. Interactions with the membrane lipids are responsible for the sensing of mechanical force for most known MS channels. Not only prokaryotic, but also eukaryotic, MS channels are able to directly sense the tension in the membrane bilayer without an additional cofactor. Sensitivity towards tension changes can be explained as result of the hydrophobic coupling between membrane and transmembrane (TM) regions of the channel. Molecular interactions of lipids with the channels may play an important role in mechanosensation. Pockets in between TM helices were identified in MscS and YnaI (TC# 1.A.23.4.3) that are filled with lipids. Fewer lipids are present in the open state of MscS than the closed. Thus, exclusion of lipid fatty acyl chains from these pockets, as a consequence of increased tension, may trigger gating. Similarly, in the eukaryotic MS channel TRAAK it was found that a lipid chain blocks the conducting path in the closed state (Rasmussen 2016).
All microsporidian genomes characterized to date contain mscS genes of both eukaryotic and bacterial origin. Berg et al. 2024 investigated the cryo-EM structure of the bacterially derived MscS2 from Nematocida displodere, an intracellular pathogen of Caenorhabditis elegans. MscS2 is the most compact MscS-like channel known and assembles into a unique superstructure in vitro with six heptameric MscS2 channels. Individual MscS2 channels are oriented in a heterogeneous manner to one another, resembling an asymmetric, flexible six-way cross joint. Microsporidian MscS2 still forms a heptameric membrane channel, but the extreme compaction suggests a new function of this MscS-like protein (Berg et al. 2024).
The generalized transport reaction proposed for MscS channels is:
osmolytes (in) and ions (in) 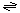 osmolytes (out) and ions (out).
osmolytes (out) and ions (out).