1.A.47 The Nucleotide-sensitive Anion-selective Channel, ICln (ICln) Family
ICln is a multifunctional protein in animals that is essential for cell volume regulation. It is found in the cytosol and is associated with the cell membrane. It is also a methylosome (a 20 S complex) subunit. It may inhibit snRNP formation. It is called the chloride channel regulator, I(Cln), the Cl- channel, nucleotide-sensitive 1A protein, and the chloride ion current-inducer protein.
ICln participates in cell volume regulation by activation of a swelling-induced Cl- conductance pathway. Ion currents are activated during regulatory volume decrease after cell swelling. ICln reconstituted in artificial bilayers forms ion channels (Ritter et al., 2003). ICln is found in many cells and tissues of the mammal, but homologues are also found in C. elegans, D. melanogster, fungi, plants and protozoans. They are usually about 200 aas long.
Cell swelling causes ICln to redistribute from the cytosol to the cell membrane. Purified ICln, added to the extracellular medium or overexpression of farnesylated ICln leads to increased ion permeability in 3T3 fibroblasts. These channels are permeable to cations, organic osmolytes, and ATP in addition to inorganic anions. Hypotonicity induces aquaporin-2 internalization and cytosol-to membrane translocation of ICln in renal cells (Tamma et al. 2007).
The activity of ICln can be blocked by Cl- channel blockers and extracellular nucleotides. They are outward rectifying and inactivated at positive potentials. Activity is dependent on pH, Ca2+ and the lipid composition of the membrane.
The reaction catalyzed by ICln is:
small molecule (in) 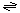 small molecule (out).
small molecule (out).