1.A.6 The Epithelial Na+ Channel (ENaC) Family
Epithelial sodium channels facilitate Na⁺ reabsorption across the apical membranes of epithelia in the distal nephron, respiratory and reproductive tracts and exocrine glands, and hence they have a role in fluid volume homeostasis, osmolarity and arterial blood pressure regulation (Enuka et al. 2012). Acid-sensing ion channels are broadly distributed in the nervous system where they contribute to sensory processes (Schuhmacher et al. 2015). ENaC family members are from animals with no recognizable homologues in other eukaryotes or bacteria. The vertebrate ENaC proteins from epithelial cells cluster tightly together on the phylogenetic tree; voltage-insensitive ENaC homologues are also found in the brain. The many sequenced C. elegans proteins, including the worm degenerins, are distantly related to the vertebrate proteins as well as to each other. At least some of these proteins form part of a mechano-transducing complex for touch sensitivity, but others function in chemosensory transduction pathways (Ben-Shahar, 2011). D. melanogaster also has many ENaC family paralogues, some closely related to each other, others very distant in sequence. Other members of the ENaC family, the acid-sensing and/or mechanosensory ion channels, ASIC1-4, are homo- or hetero-oligomeric neuronal Zn2+ and H+-gated, mechanosensitive channels that mediate pain sensation in response to tissue acidosis. Two extracellular histidines (his-162 and his-339) potentiate Zn2+ activation while another (his-72) mediates pH sensitivity (Baron et al., 2001). ASIC1-4 also mediate light touch sensation and are excited by hair movement. The homologous Helix aspersa (FMRF-amide)-activated Na+ channel is the first peptide neurotransmitter-gated ionotropic receptor to be sequenced. Salty taste is mediated by an ENaC channel in the fungiform papillae in the dorsal epithelium of the anterior tongue. Activation of acid-sensing ion channel 1a (ASIC1a) occurs in response to surface trafficking (Chai et al., 2010). The stress response protein, SERP1, regulates ENaC biogenesis (Faria et al., 2012). Potassium activates mTORC2-dependent SGK1 phosphorylation to stimulate ENaC (Saha et al. 2023). The epithelial sodium channel is a drug target (Lemmens-Gruber and Tzotzos 2023). The optimization of electrochemical immunosensors can be used to detect epithelial sodium channel as a biomarker of hypertension (Lestari et al. 2023). 6-Iodoamiloride is an amiloride analog, a potent inhibitor of ASICs (Finol-Urdaneta et al. 2023). The intricate interplay between proteases and ENaC emphasizs the significance of proteolytic activation in maintaining sodium and fluid balance in both health and disease (Aufy et al. 2023).
Epithelial Na+ channel (ENaC)/degenerin family members are involved in mechanosensation, blood pressure control, pain sensation, and the expression of fear. They display a form of desensitization (Roy et al. 2013). Members all exhibit the same apparent topology, each with N- and C-termini on the inside of the cell, two amphipathic transmembrane spanning segments, M1 and M2, and a large extracellular loop (Saugstad et al. 2004). The extracellular domains contain numerous highly conserved cysteine residues. They are proposed to serve a receptor function. Welsh et al. (2002) presented three models whereby members of the ENaC family sense mechanostimulation. Their preferred model involves tethering the channel protein to extracellular matrix proteins such as collagens and/or intracellular cystoskeletal proteins such as α- and β-tubulins. Carnally et al., 2008 have presented evidence, based on an X-ray crystal structure, that ASIC1a assembles as a heterotrimer. Carattino (2011) has reviewed the structural mechanisms underlying the functions of epithelial sodium channel/acid-sensing ion channels. Opening of the ion conductive pathway involves coordinated rotation of the second transmembrane-spanning domains (Tolino et al., 2011). The second TMS modulates channel gating in response to shear stress (Abi-Antoun et al., 2011). ASIC- and ENaC-types of Na+ channels exhibit different conformational changes (Hanukoglu 2016). The ion selectivity filter has been discussed (Hanukoglu 2016). Interactions between the epithelial sodium channel gamma-subunit and claudin-8 modulates paracellular sodium permeability in the renal collecting duct (Sassi et al. 2020). Reactive species generated by heme impair alveolar epithelial sodium channel function in acute respiratory distress syndrome (Aggarwal et al. 2020). DEG/ENaC/ASIC channels vary in their sensitivities to anti-hypertensive and non-steroidal anti-inflammatory drugs (Fechner et al. 2021). Epithelial sodium channels exhibit sodium self-inhibition (Lawong et al. 2023).
Mammalian ENaC is important for the maintenance of Na+ balance and the regulation of blood pressure. Three homologous ENaC subunits, α, β and γ, have been shown to assemble to form the highly Na+-selective channel. Only the dehydrated form of Na+ (or Li+) is transported. The stoichiometry of the three subunits is αβγ in a heterotrimeric architecture, and they form a triangular pyramid-shaped funnel (Edelheit et al. 2014). A structural model has been proposed in which the properties of the channel are conferred by the second TMS together with the preceding hydrophobic region that may loop into the membrane as do the P-regions of VIC family members. The selectivity filter of the epithelial Na+ channel α-subunit is at least in part determined by residues Ser580 to Ser592 following the second TMS. Residues conferring cation selectivity are in both M2 and the preceding loop. Negatively charged residues in M2 of the mammalian α-subunit are important, as two substitutions, αE595C and αD602C confer K+ permeability (Sheng et al., 2001b). ASICs, proton-gated cation channels, contribute to fast synaptic transmission and have roles in fear conditioning and nociception. Apart from activation at low pH, ASIC1a also undergoes several types of desensitization, including acute desensitization, which terminates activation; steady-state desensitization, which occurs at sub-activating proton concentrations and limits subsequent activation; and tachyphylaxis, which results in a progressive decrease in response during a series of activations (Holm et al. 2024).
The C-terminus of each ENaC subunit contains a PPXY motif which when mutated or deleted in either the β- or γ-ENaC subunit leads to Liddle's syndrome, a human autosomal dominant form of hypertension. In this disease, the mutation induces abnormally high levels of channel expression due to a loss of interaction with the inhibitory Nedd4 protein. Nedd4 regulates the activity of the epithelial Na+ channel in normal people but not in those suffering from Liddle's syndrome. Multiple WW domains in Nedd4 mediate the interaction with all three subunits of ENaC, α, β and γ, and WW domains 2-4 are most important for this interaction (Snyder et al., 2001). Cys palmitoylation of the β subunit modulates gating of the epithelial sodium channel (Mueller et al., 2010). Bile acids, especially tauro-deoxycholic acid (t-DCA), modify the function of the acid-sensing ion channel ASIC1a and other members of the epithelial sodium channel (ENaC)/degenerin (DEG) ion channel family (Ilyaskin et al. 2019). Polyunsaturated fatty acids (PUFAs) and their analogs modulate the activities of diverse ion channels, and these lipids potentiate acid-sensing ion channels (ASICs), leading to increased activity (Bandarupalli et al. 2024). Functional expression of the proton sensors ASIC1a, TMEM206, and OGR1 together with BKCa channels is associated with cell volume changes and cell death under strongly acidic conditions (Pissas et al. 2024).
Cystic fibrosis (CF) lung disease is caused by the loss of function of the cystic fibrosis transmembrane conductance regulator (CFTR) combined with hyperactivation of the epithelial sodium channel (ENaC). In the lung, ENaC is responsible for movement of sodium. Hyperactivation of ENaC, which creates an osmotic gradient that pulls fluid out of the airway, contributes to reduced airway hydration, causing mucus dehydration, decreased mucociliary clearance, and recurrent acute bacterial infections. ENaC represents a therapeutic target to treat patients with CF independently of their underlying CFTR mutation. SPX-101, a peptide resulting from proteolytic digestion of SPLUNC1 (Q9NP55; 256 aas) binds selectively to ENaC and promotes internalization of the α-, β-, and γ-subunits. Removing ENaC from the membrane with SPX-101 causes a significant decrease in amiloride-sensitive current, promting survial of CF patients (Scott et al. 2017).
Acid-sensing ion channels (ASICs) have been implicated in perception of pain, ischaemic stroke, mechanosensation, learning and memory. They are implicated in touch, pain, digestive function, baroreception, blood volume control and hearing (Chen and Wong 2013). Jasti et al. (2007) reported the low-pH crystal structure of a chicken ASIC1 deletion mutant at 1.9 Å resolution. Each subunit of the chalice-shaped homotrimer is composed of short amino and carboxy termini, and two transmembrane helices. A bound chloride ion is present. A disulphide-rich, multidomain extracellular region is enriched in acidic residues with carboxyl-carboxylate pairs, suggesting that at least one carboxyl group bears a proton. Electrophysiological studies on aspartate-to-asparagine mutants confirmed that these carboxyl-carboxylate pairs participate in proton sensing. Between the acidic residues and the transmembrane pore lies a disulphide-rich 'thumb' domain poised to couple the binding of protons to the opening of the ion channel. The results demonstrated that proton activation involves long-range conformational changes. The Akt and Sgk protein kinases are components of an insulin signaling pathway that increases Na+ absorption by up-regulating membrane expression of ENaC via a regulatory system that involves inhibition of Nedd4-2 (Lee et al., 2007).
Gonzales et al. (2009) presented the structure of a functional acid-sensing ion channel in a desensitized state at 3 Å resolution, the location and composition of the approximately 8 Å thick desensitization gate, and the trigonal antiprism coordination of caesium ions bound in the extracellular vestibule. Comparison of the acid-sensing ion channel structure with the ATP-gated P2X(4) receptor revealed similarity in pore architecture and aqueous vestibules, suggesting that there are unanticipated yet common structural and mechanistic principles (Gonzales et al., 2009). ENaCs have been used to form solid-state nanopores with diameters in the range of 150-200 nm and a thickness <1 micron which could serve as a platform to enhance the throughput of ion-channel characterization using Black Lipid Membranes (Khan et al. 2016). Acid-sensing ion channels (ASICs) are weakly sodium selective (sodium:potassium ratio approximately 10:1), while ENaCs show a high preference for sodium over potassium (>500:1). The pre-TMS1 and TMS1 regions of mASIC1a channels are major determinants of ion selectivity (Sheikh et al. 2021).
The activity of the epithelial sodium channel (ENaC) is modulated by multiple external factors, including proteases, cations, anions and shear stress. The resolved crystal structure of acid-sensing ion channel 1 (ASIC1), and mutagenesis studies suggest that the large extracellular region is involved in recognizing external signals that regulate channel gating. The thumb domain in the extracellular region of ASIC1 has a cylinder-like structure with a loop at its base that is in proximity to the tract connecting the extracellular region to the transmembrane domains. This loop has been proposed to have a role in transmitting proton-induced conformational changes within the extracellular region to the gate. Shi et al. (2011) examined whether loops at the base of the thumb domains within ENaC subunits have a similar role in transmitting conformational changes induced by external Na+ and shear stress. Mutations at selected sites within this loop in each of the subunits altered channel responses to both external Na+ and shear stress. The most robust changes were observed at the site adjacent to a conserved Tyr residue. In the context of channels that have a low open probability due to retention of an inhibitory tract, mutations in the loop activated channels in a subunit-specific manner. This loop may have a role in modulating channel gating in response to external stimuli, consistent with the hypothesis that external signals trigger movements within the extracellular regions of ENaC subunits that are transmitted to the channel gate (Shi et al., 2011).
As noted above, epithelial sodium channels (ENaC) consist of three homologous subunits. Channels composed solely of alpha and beta subunits (αβ-channels) exhibit a very high open probability (Po) and reduced sensitivity to amiloride, in contrast to channels composed of alpha and gamma subunits or of all three subunits (i.e., αγ- and αβγ-channels). A mutant channel comprised of alpha and beta subunits, and a chimeric gamma subunit where the region immediately preceding (beta12 and wrist) and encompassing the second transmembrane domain (TMS2) has been replaced with the corresponding region of the beta subunit (gamma-betaTMS2) and showed characteristics reminiscent of αβ-channels, including a reduced potency of amiloride block and a loss of Na+ self-inhibition (reflecting an increased Po) (Shi and Kleyman 2013). Substitutions at key pore-lining residues of the γβ-TMS2 chimera enhanced the Na+ self-inhibition response, whereas key γ-subunit substitutions reduced the response. Furthermore, multiple sites within the TMS2 domain of the γ-subunit were required to confer high amiloride potency. Thus, pore-lining residues in the γ-subunit are important for proper channel gating and its interaction with amiloride.
Acid-sensing ion channels (ASICs) are cation selective proton-gated channels expressed in neurons that participate in diverse physiological processes including nociception, synaptic plasticity, learning, and memory. ASIC subunits contain intracellular N- and C- termini, two transmembrane domains that constitute the pore and a large extracellular loop with defined domains termed the finger, beta-ball, thumb, palm, and knuckle. Krauson and Carattino 2016 examined the contribution of the finger, beta-ball and thumb domains to activation and desensitization. The beta-ball and thumb domains reside apart in the resting state, but they become closer to each other in response to extracellular acidification. The thumb domain probably moves upon continuous exposure to an acidic extracellular milieu assisting with the closing of the pore during channel desensitization.
The ENaC Family has been reviewed by Hanukoglu and Hanukoglu 2016. ENaC dependent reabsorption of Na in kidney tubules regulates extracellular fluid (ECF) volume and blood pressure by modulating osmolarity. In multi-ciliated cells, ENaC is located in cilia and plays an essential role in the regulation of epithelial surface liquid volume necessary for cilial transport of mucus and gametes in the respiratory and reproductive tracts, respectively. The subunits that form ENaC (named as alpha, beta, gamma and delta, encoded by genes SCNN1A, SCNN1B, SCNN1G, and SCNN1D) are in the ENaC/Degenerin superfamily. The earliest appearance of ENaC orthologs is in the genomes of the most ancient vertebrate taxon, Cyclostomata (jawless vertebrates) including lampreys, followed by earliest representatives of Gnathostomata (jawed vertebrates) including cartilaginous sharks. Among Euteleostomi (bony vertebrates), Actinopterygii (ray finned-fishes) branch has lost ENaC genes. Yet, most animals in the Sarcopterygii (lobe-finned fish) branch including Tetrapoda, amphibians and amniotes (lizards, crocodiles, birds, and mammals), have four ENaC paralogs (Hanukoglu and Hanukoglu 2016).
ENaC subunits are subject to numerous posttranslational modifications, including glycosylation, protease activation, disulfide bond formation and palmitoylation, each of which modulates channel function. For example, glycan addition is regulated by sodium and affects protease activation at the cell surface, protein trafficking, sodium-dependent regulation, and sodium transport. Glycosylation of the alpha subunit also determines whether a chaperone, Lhs1/GRP170, selects the protein for endoplasmic reticulum-associated degradation. Recognition by this chaperone is blocked by assembly of the ENaC transmembrane domains. In contrast, cytosolic lysines are acetylated in the early secretory pathway, which inhibits ubiquitination and endocytosis at the cell surface (Buck and Brodsky 2018).
ENaC exhibits a very high selectivity for Na+ over other cations, including K+, and this selectivity greatly exceeds that of the closely related acid-sensing channels (ASICs). Yang and Palmer 2018 assessed the roles of two regions of the ENaC transmembrane pore in the determination of cation selectivity. Mutations of conserved amino acids with acidic side chains near the cytoplasmic end of the pore diminish macroscopic currents but do not decrease the selectivity of the channel for Na+ versus K+. In the WT channel, voltage-dependent block of Na+ currents by K+ or guanidinium+, neither of which have detectable conductance, suggested that these ions permeate only approximately 20% of the transmembrane electric field. The site of K+ block appears to be nearer the extracellular end of the pore, close to a putative selectivity filter, but.while this region affects the Li:Na selectivity, the high Na:K selectivity was maintained. Yang and Palmer 2018 concluded that a different part of the pore constitutes the selectivity filter in ENaC versus ASIC.
ENaC regulates Na+ and water homeostasis. These heterotrimeric channels harbor protease-sensitive domains critical for gating. Noreng et al. 2018 presented the structure of human ENaC in the uncleaved state as determined by single-particle cryo-EM. The ion channel is composed of a large extracellular domain and a narrow transmembrane domain. ENaC assembles with a 1:1:1 stoichiometry of alpha:beta:gamma subunits arranged in a counter-clockwise manner. The shape of each subunit is reminiscent of a hand with key gating domains of a 'finger' and a 'thumb'. Wedged between these domains is the protease-sensitive inhibitory domain poised to regulate conformational changes of the 'finger' and 'thumb'.
Despite the sequence homology between acid-sensing ion channels (ASICs) and epithelial sodium channel (ENaCs), these channels display very different functional characteristics. Whereas ASICs are gated by protons and show a relatively low degree of selectivity for sodium over potassium, ENaCs are constitutively active and display a remarkably high degree of sodium selectivity. Differences in the transmembrane domains of these ion channels help explain some of their distinct functional properties (Kasimova et al. 2019). Enhanced glycolysis causes extracellular acidification and activates acid-sensing ion channel 1a in hypoxic pulmonary hypertension (Tuineau et al. 2024). Novel ENaC inhibitors for direct delivery to the airways via inhalation have been developed. These ENaC inhibitors have the pyrazine core of first-generation inhibitors replaced with a pyrrolopyrazine. Aiming for high retention at the surface of the lung following inhalation, optimisation of this template focused on significantly increasing polarity to minimize passive cellular permeability. The resulting optimized clinical candidate ETD001 demonstrated potent inhibition of ENaC (59 nM) prolonged retention in the airways of rats (13 % of the delivered dose retained after 6h) following intratracheal administration and a potent and long-acting effect in a sheep model of mucociliary clearance following inhalation (ED100 (4-6h) = 9 μg/kg). ETD001 entered a phase II study in CF patients in July 2024 (Danahay et al. 2025).
The generalized transport reaction for Na+ channels is:
Na+ (out) 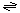 Na+ (in).
Na+ (in).
That for the degenerins is:
Cation (out) 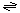 cation (in).
cation (in).