9.A.11 The Dipicolinic Acid Transporter (DPA-T) Family
The products of the hexacistronic spoVA operon (spoVAA-F) of Bacillus subtilis is involved in the uptake of dipicolinic acid (DPA) into the forespore during sporulation and its release from the mature spore during germination (Paredes-Sabja et al., 2008). SpoVAA and SpoVAD have both been implicated in transport and shown to be integral membrane proteins (Vepachedu and Setlow, 2005). Homologues of these six proteins are found in other Bacillus species as well as in Geobacter kaustophilus and Oceanobacillus iheyensis. SpoVAD has homologues (or partial homologues) in many other bacteria as well. Moreover, SpoVAD and SpoVAC are homologous in their C-terminal domains, and the N-terminal domain of SpoVC (323 aas) is homologous to most of SpoVE (150 aas). SpoVAF is homologous to GerA1 (482 aas; 6 TMS) (P07868). It is not yet clear that all the proteins encoded within the spoVA operon are constituents of the transporter.
The generalized reaction believed to be catalyzed in the spoVA operon protein is:
DPAin 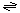 DPAout
DPAout
The release of dipicolinic acid (DPA) during the germination of Bacillus subtilis spores by the cationic surfactant dodecylamine exhibits a pH optimum of ~9 and a temperature optimum of 60oC. DPA release during dodecylamine germination with fourfold-elavated levels of the SpoVA proteins was about fourfold faster than DPA release during dodecylamine germination of wild-type spores and was inhibited by HgCl2 (Ramana et al., 2007). Spores carrying temperature-sensitive mutants in the spoVA operon were also temperature sensitive in DPA release during dodecylamine germination as well as in lysozyme germination of decoated spores. In addition to DPA, dodecylamine triggered the release of amounts of Ca2+ almost equivalent to those of DPA, and at least one other abundant spore small molecule, glutamic acid, was released in parallel with Ca2+ and DPA. These data indicate that (i) dodecylamine triggers spore germination by opening a channel in the inner membrane for Ca2+-DPA and other small molecules, (ii) this channel is composed at least in part of proteins, and (iii) SpoVA proteins are involved in the release of Ca2+-DPA and other small molecules during spore germination, perhaps by being a part of a channel in the spore's inner membrane (Ramana et al., 2007).