1.C.75 The Serratia-type Pore-forming Toxin (S-PFT) Family
The Serratia marcescens hemolysin, ShlA, represents the prototype of a growing family of pore-forming toxins. Although only S. marcescens hemolysin has been studied in great molecular detail, available bacterial genome sequences reveal Serratia hemolysin homologues in additional species. This family of toxins has little in common with the pore forming toxins of E. coli type (RTX toxins), the Staphylococcus aureus α-toxin or the thiol activated toxin of group A β-hemolytic streptococci (Streptolysin O) (Hertle, 2005). Studies on erythrocytes, eukaryotic cells and artificial black lipid membranes, have shown that the mechanism of pore formation by ShlA is different from other pore-forming toxins. The S. marcescens hemolysin proteins ShlB and ShlA, exhibit homologues in Proteus mirabilis, Haemophilus ducreyi, Yersinia pestis, Yersinia enterocolitica, Edwardsiella tarda, Photorhabdus luminescens and Xylella fastidiosa, all γ-proteobacteria. The Serratia protein contains an N-terminal haemagglutination activity domain, suggested to be a carbohydrate-dependent haemagglutination site found in a range of haemagglutinins and
haemolysins. It also contains at least 5 central and C-terminal haemagglutination repeats.
The family of Serratia type pore-forming toxins are exported by two partner secretion (TPS; TC #1.B.20). However, ShlA shows greater sequence similarity to regions of members of the autotransporter (AT) proteins, even though ShlB transports ShlA across the outer Serratia membrane (TC #1.B.20.1.1). In fact, residues 40-826 of the ShlA (1608 aas) are homologous to residues 72-896 of the AidA-1 precursor of E. coli (TC #1.B.12.1.1), but residues 962-1256 in ShlA are also homologous to residues 178-455 in AidA-1, and residues 155-318 in ShlA are homologous to residues 26-209 in AidA-1, suggesting the presence of repeat elements. Moreover, ShlA shows significant sequence similarity with the RTX cytotoxin of Vibrio vulnificus in the Clostridial Cytotoxin (CCT) family (TC #1.C.57.3.4). Residues 965-1196 show similarity with the latter residues 17-237, and residues 1180-1396 in ShlA show similarity with the latter residues 57-285, again suggesting the presence of repeat units in ShlA. Our analyses suggest that the S-PFT family is distantly related to both the RTX-toxin (TC #1.C.11) and the CCT (TC #1.C.57) families.
Not only Serratia type pore-forming toxins, but also adhesins from Bordetella pertussis, Erwinia chrysanthemi and Haemophilus influenzae are secreted via TPS systems. The uniqueness of the family is underlined by the fact that activation of ShlA by ShlB strictly requires phosphatidylethanolamine as a cofactor. ShlA undergoes a conformational change during activation (Hertle, 2005).
As an insect pathogen, Xenorhabdus nematophila possesses an arsenal of virulence factors, one of which is XhlA, required for full virulence towards Manduca sexta larvae. Lrp (leucine-responsive regulatory protein), FlhDC (master regulator of flagella synthesis), and iron (II) limitation positively influenced xhlA transcript levels, suggesting that XhlA is linked to nutrient acquisition and motility. Cowles and Goodrich-Blair 2005 examined its cellular targets and found that XhlA is a cell-surface associated haemolysin that lysed the two most prevalent types of insect immune cells (granulocytes and plasmatocytes) as well as rabbit and horse erythrocytes. Thus, the need for xhlA for full virulence and XhlA activity towards insect immune cells suggests that this haemolysin functions in immune evasion during infection.
The reaction catalyzed by the ShlA Serratia toxin and other homologous toxins is:
small molecule (in) 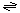 small molecule (out)
small molecule (out)